NUS Medicine researchers create AI-guided gene-editing tool for more precise and safer DNA correction
Published: 18 Mar 2026

From left to right: Assistant Professor Jungjoon K. Lee, Department of Biochemistry, Synthetic Biology for Clinical and Technological Innovation (SynCTI), Synthetic Biology Translational Research Programme (TRP), NUS Medicine; Dr Gao Ni, Research Fellow, Department of Biochemistry, NUS Medicine; and Mr Matthias Lim Boon Han, Student, NUS Medicine
Researchers at NUS Medicine have developed a revolutionary new method to improve compact gene-editing tools known as base editors, which enable smaller, more precise DNA correction tools that may be safer for future gene therapies.
Published in Advanced Science, the study was led by Assistant Professor Jungjoon K. Lee, from the Department of Biochemistry, Synthetic Biology for Clinical and Technological Innovation (SynCTI) and Synthetic Biology Translational Research Programme (TRP) at NUS Medicine. The team combined artificial intelligence-driven protein modelling with a novel bacterial evolution platform to engineer enhanced versions of SsdAtox, a compact DNA-editing enzyme. The optimised variants matched or exceeded the performance of leading editors such as BE4max at multiple human gene targets, while reducing unwanted DNA damage and cellular toxicity.
Base editors are powerful tools that can correct single-letter mistakes in DNA — the type of mutations that cause many inherited diseases — without cutting the DNA completely. This makes them potentially safer than earlier gene-editing technologies. However, many high-performing base editors are large, making them difficult to package into delivery vehicles such as adeno-associated viruses (AAVs), which are commonly used in gene therapy. In addition, increasing editing efficiency can also increase the risk of unintended DNA changes.
SsdAtox is about two-thirds the size of commonly used base editors, making it attractive for therapeutic delivery. But in its natural form, it edits DNA inefficiently and can cause unwanted DNA damage and toxicity. The team’s goal was to improve its performance while keeping it compact.
“In gene editing, improving efficiency often increases the risk of unwanted DNA damage,” Asst Prof Lee said. “By combining AI-guided structural modelling with a three-layer screening system, we were able to improve activity while reducing those side effects. Beyond this specific enzyme, our approach provides a general strategy for developing safer and more precise gene-editing tools.”
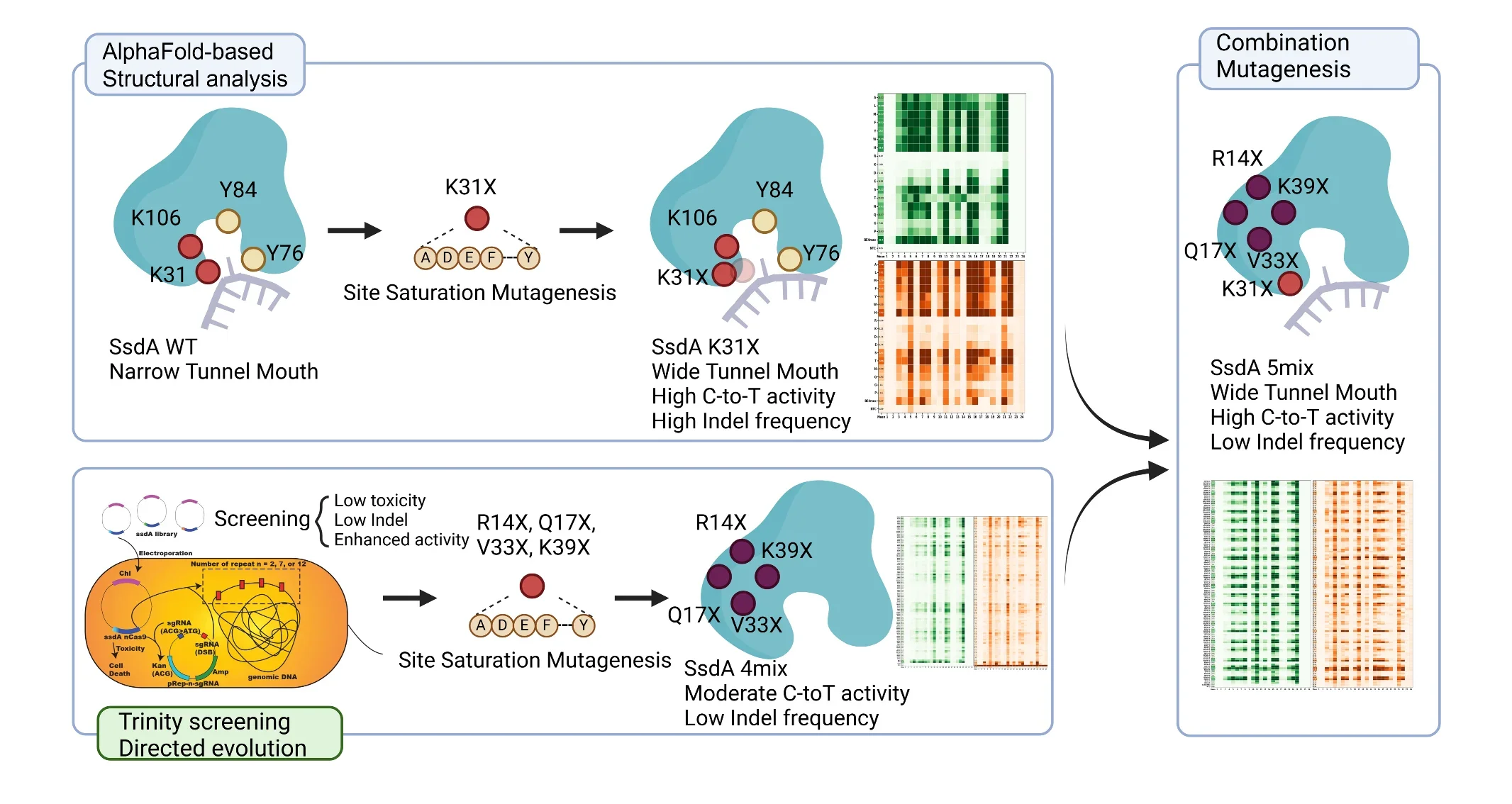
AlphaFold-based structural analysis.
The team used a two-step strategy to achieve this. First, they used AlphaFold3, an advanced AI system that predicts protein structures, to identify a key part of the enzyme that controls how DNA enters its active site. By changing a specific position in the enzyme (known as K31), they were able to widen this “entrance,” allowing the enzyme to work more efficiently.
Second, they developed a new screening platform called Trinity-Screen. This system tests three things at the same time inside bacterial cells: does the enzyme edit DNA efficiently; does it avoid causing harmful DNA breaks; and is it non-toxic to cells?
Only enzyme variants that checked all three criteria survived repeated rounds of selection. The most promising mutations were then combined and tested in human cells at 24 artificial DNA targets and 10 natural human gene sites. The best-performing engineered variants showed:
- Up to 11.8 times higher editing efficiency compared to the original SsdAtox
- About half the unwanted DNA break rate compared to earlier high-activity mutants
- Ten times lower toxicity in bacterial cells
- Up to 31-fold overall improvement, measured using a newly developed scoring system
Among the top variants, one reduced unwanted DNA breaks by 37% compared to BE4max at multiple gene targets. Importantly, the improved enzymes also edited DNA within a narrower and more consistent “editing window,” meaning their activity may be more predictable across different genes. To fairly compare different gene-editing tools, the team developed a new scoring system called the Base Editor Performance Index (BEPI). Instead of looking only at editing efficiency, BEPI balances how well the enzyme edits DNA against how much unwanted damage it causes, which provides a clearer overall picture of performance. As the optimised SsdAtox variants are smaller than many existing editors, they may be easier to package into viral delivery systems used in gene therapy. This could expand treatment possibilities for genetic diseases that are currently difficult to target.
The study also demonstrates one of the first confirmed examples of using AlphaFold3-guided protein modelling to directly improve the performance of a genome-editing enzyme.
“By combining AI-driven design with a powerful screening system, we hope that this framework can help accelerate the development of safer gene-editing tools more broadly,” added Asst Prof Lee.
View the press release here.


