AI designs drug combinations specifically for women or men to optimise heart valve disease treatment
Published: 11 Jun 2025

From left: Members of the research team, Prof Dean Ho, Dr Peter Wang, Brian Aguado
Aortic valve stenosis (AVS), which affects nearly 1 in 8 adults over the age of 75 globally, is a serious heart condition that exhibits sex-specific responses to treatment. It occurs when the aortic valve—which controls blood flow from the heart to the rest of the body—becomes narrowed or stiff, making it harder for blood to flow efficiently. It can cause symptoms like chest pain, fatigue, shortness of breath, or fainting. Left untreated, severe AVS may progress to heart failure.
Harnessing the artificial intelligence (AI) driven platform, IDentif.AI, researchers from the Institute for Digital Medicine (WisDM), Yong Loo Lin School of Medicine, National University of Singapore (NUS Medicine), Cancer Science Institute of Singapore, NUS and the Shu Chien-Gene Lay Department of Bioengineering at the University of California San Diego, sought to uncover sex-specific drug combinations that may slow or halt AVS progression. IDentif.AI optimised drug combinations to inhibit aortic valve myofibroblast activation—a hallmark of AVS which refers to a process causing valve cells to stiffen and scar—in female and male valve cells isolated from laboratory models. The AI platform pinpointed a list of female-biased combinations that were more effective in addressing AVS in female cells, while AI-optimised male-biased combinations were notably more effective in male cells.
Professor Dean Ho, Director of WisDM, NUS Medicine, who co-led the study with Professor Brian Aguado at the University of California San Diego, said, “Our collaborative study demonstrates that men and women may require different medications or drug combinations to achieve the best outcomes in diseases like AVS. By optimising AI and hydrogel biomaterials, we can quickly identify and validate personalised therapies that consider these key differences. Beyond laying the groundwork for advancing AVS-specific treatment strategies, our study highlights the importance of considering sex as a biological variable and possible disparities in treatment outcomes.”
Published in Science Advances, the research team selected eight drug candidates that target pathways of aortic valve myofibroblast activation. A minimum of 12 samples of male and female valvular interstitial cells (VICs)—the key cells that maintain heart valve structure and function—were isolated directly from laboratory models and cultured in hydrogel biomaterials that closely mimicked the stiffness and environment of healthy and diseased human heart valves. The team then tested 59 drug combinations in both female and male VICs cultured in hydrogel biomaterials and determined their efficacy by their respective inhibition levels of the myofibroblast activation, a process which causes valve cells to stiffen and scar. The results were then collated on the IDentif.AI platform, which ranked the combinations and pinpointed the most effective sex-specific drug cocktails for both male and female VICs.
Dr Peter Wang, co-author of the study, from Prof Ho’s research team at WisDM, NUS Medicine, added, “With the findings from our study, we aim to accelerate the development of sex-specific drug combinations for diseases like AVS and emphasise the importance of considering sex as a biological variable in treatment design.” Dr Wang is also from the NUS N.1 Institute for Health and the Department of Biomedical Engineering at NUS CDE.
Prof Aguado added, “The hydrogel biomaterials developed in our laboratory enabled the discovery that male and female VICs have sex-dependent synergistic responses to drug combinations. Our observations would not have been possible using conventional tissue culture plastic materials, revealing the importance of the cell culture microenvironment to discover sex-specific biological mechanisms and paths to precision treatments. We look forward to continued work using lab-grown humanised in vitro cell culture models of valve disease as a next step to accelerate drug combination optimisation and clinical trial design.”
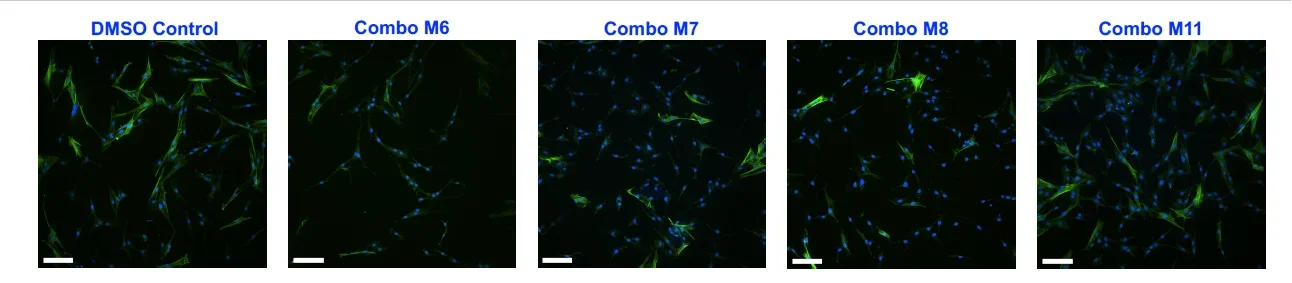 In Figure 1, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for male VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (M6, M7, and M8) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (M11).
In Figure 1, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for male VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (M6, M7, and M8) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (M11).
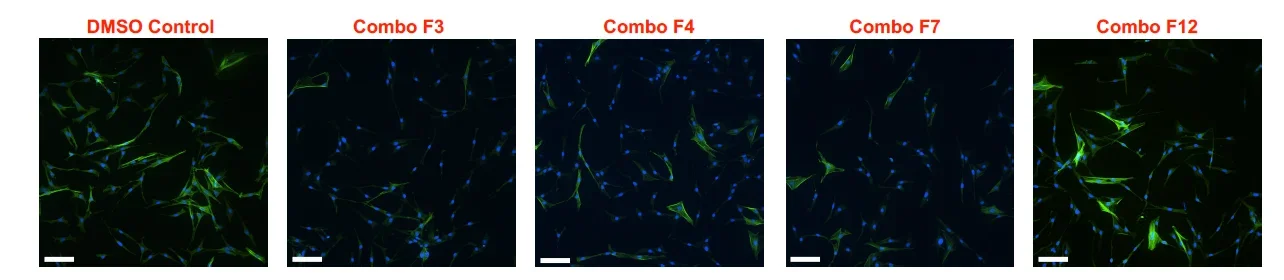 In Figure 2, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for female VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (F3, F4, and F7) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (F12).
In Figure 2, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for female VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (F3, F4, and F7) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (F12).
Read more in the press release here.


 In Figure 1, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for male VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (M6, M7, and M8) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (M11).
In Figure 1, the immunofluorescent images representing the inhibition of myofibroblast activation are shown for male VICs cultured with IDentif.AI-pinpointed effective and ineffective drug combinations. The inhibition of myofibroblast activation is represented by the reduction of alpha-smooth muscle actin (αSMA), which are stained in green. The cell nuclei are stained in blue to differentiate from αSMA. The staining of VICs treated with effective drug combinations (M6, M7, and M8) resulted in less presence of αSMA (green), while more staining of αSMA (green) can be seen when treated with the ineffective combination (M11). 