Neurobiology
Chapter 1: Sensation and Perception, & Fundamentals of Sensory System
Asst Prof Shuo-Chien Ling, Ph.D.
Laboratory of Molecular Neurodegeneration
NUS Yong Loo Lin School of Medicine Department of Physiology
Email: phsling@nus.edu.sg

Introduction

Have you ever wondered how we sense and perceive the world - how the eyes see; the skin detect cold, heat, pain; the ears recognise certain sounds; the nose identify smells; and the mouth enjoy different tastes?
In this e-Book, you will learn the science behind our senses and perception. Sensory neurophysiology will help you understand how we sense and perceive the world.
Overview
In this chapter, we will first focus on the types and functions of sensory system and the principles of organisation and processing of sensory information. We will also cover three special senses: taste, smell and hearing.
Learning Objectives
At the end of this chapter, you should be able to:
-
1Identify the types and explain the functions of sensory systems2Explain the principles of organisation and processing of sensory information
1.1. Functions of Sensory Systems
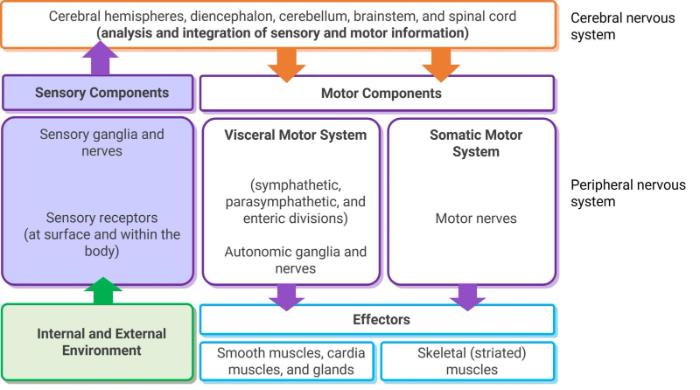
The function of each sensory system is to provide the central nervous system (CNS) with a representation of external world.

The Greek philosopher Aristotle defined the five senses.
We perceive sensory signals when they reach a level of conscious awareness.
Stimulus that usually do not reach conscious awareness include changes in muscle stretch and tension as well as a variety of internal parameters that the body monitors to maintain homeostasis such as blood pressure and pH. These processes are important in maintaining physiological homeostasis.
Hence, we will primarily consider sensory stimuli whose processing reaches the conscious level of perception.
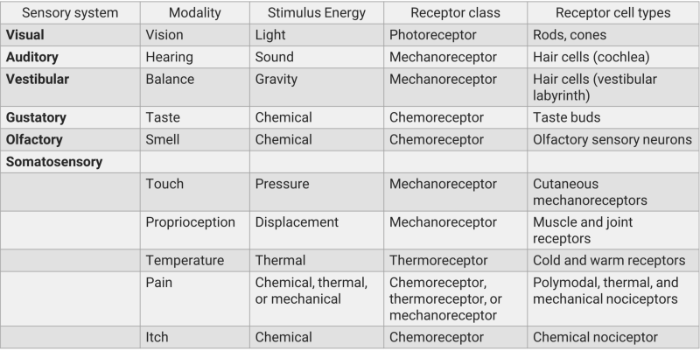
These stimuli are those associated with the special senses of vision, hearing, taste, smell and equilibrium, and the somatic senses of touch, temperature, pain, itch and proprioception. Proprioception, which is defined as the awareness of body movement and position in space, is mediated by muscle and joint sensory receptors call proprioceptors and may be either subconscious or conscious.

If you close your eyes and raise your arm above your head, you are aware of its position because of the activation of proprioceptors.
Sensory systems bring the information to an individual.
Video 1: Sensory Systems
Watch the video below for more information.
1.2. General Properties of Sensory Systems
How does the nervous system encode and process sensory stimuli?
Let's first consider the general properties of sensory pathways. Here, we will take a look at unique receptors and pathways that distinguish the different sensory systems from one another and use three special senses, taste, smell and hearing as examples.

Imagine that you are in the wild.
What do you need to do to survive?
Is there a lion or tiger hiding in the grass ready to eat you?
You need to see, hear, smell, and feel. How does the nervous system do that?
In essence, the sensory system needs to communicate the key features of any stimuli: what is the stimuli, where, how strong it is (intensity), the duration (for how long the stimuli is on). The type of stimuli, we call sensory modality, are vision, hearing, hearing, etc.
All sensory pathways have certain elements in common. They begin with a stimulus, in the form of physical energy that acts on a sensory receptor. The receptor is a transducer that converts the stimulus into an intra-cellular signal, usually, a change in membrane potential. If the stimulus is above threshold, action potentials pass along a sensory neuron to the CNS, where incoming signals are integrated. At each synapse along the pathway, the nervous system can modulate and shape the sensory information.
Key Concepts
-
- Coding and processing distinguish stimulus properties
-
-
- Sensory modality (what?)
- Location of the stimulus (where?)
- Intensity (how much?)
- Duration/timing (how long?)
-
-
- Receptors are sensitive to particular forms of energy.
- Sensory transduction converts stimuli into graded potentials.
- A sensory neuron has a receptive field.
-
-
- Lateral inhibition
-
-
- The CNS integrates sensory information.
-
-
- Labeled line
-
1.3. Sensory Receptors
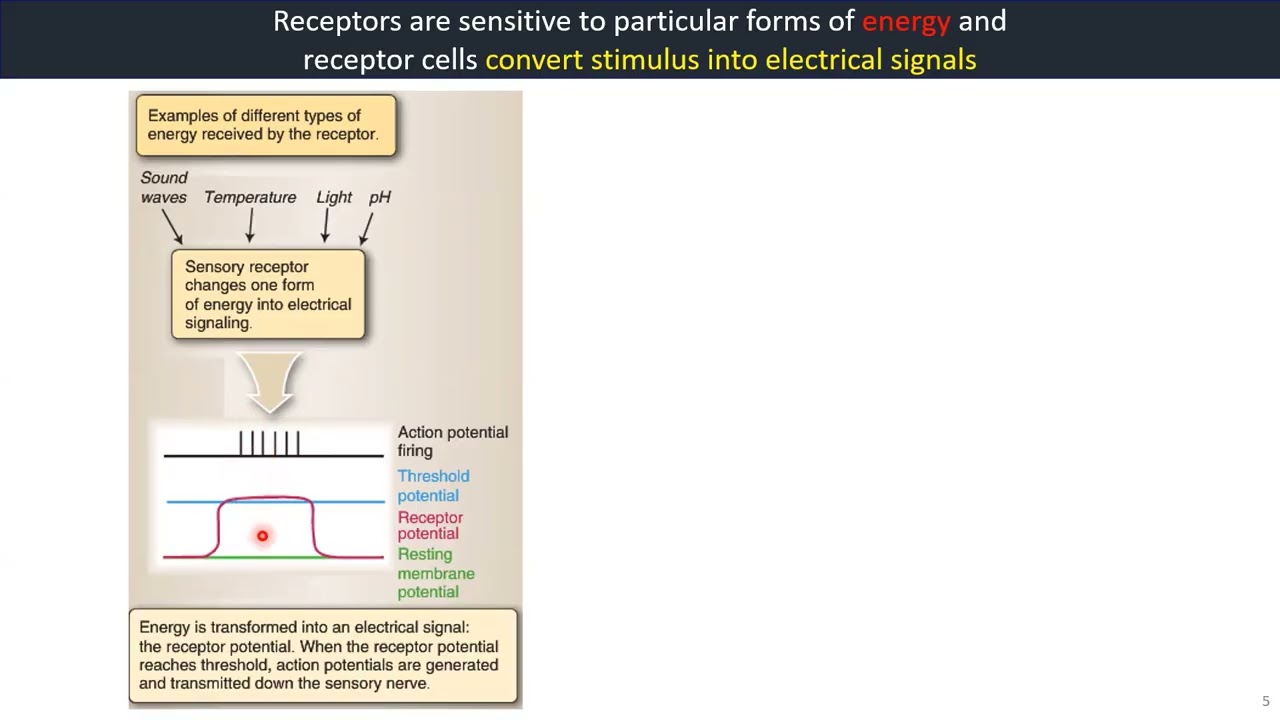
Receptors are sensitive to particular forms of energy and receptor cells convert stimulus into electrical signals.
Video 2: Sensory Receptors
Sensory receptors detect information from the environment, such as light and sound, or from our own bodies, such as touch and body position.
Watch the video below for more information.
Reference: (1) Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, Figure 3.5; (2) Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 8.11
Sensory receptors act as transducers in that they transform a physical or chemical stimulus (or form of energy) into an electrical impulse. They are specialised to detect sensory information and translate stimuli into receptor potentials, or electrical signaling within the receptor, caused by the opening and closing of ion channels. Each sensory receptor has a receptive field, which lets us discriminate the location of the sensory stimulus.
Receptor potentials, also referred to as generator potentials, are electrical impulses transduced by the sensory receptor. The receptor potential is a graded response that depends on the magnitude of the sensory stimulus and encodes for duration and intensity. Because a receptor potential dissipates after a few millimeters, an action potential must be generated to travel the long distance between the sensory receptor and the CNS. In order to generate an action potential, the depolarisation of the membrane at the sensory receptor must reach threshold. The firing frequency of the action potential in the sensory nerve is modulated by the receptor potential: The greater the stimulus, the greater the receptor potential, and the higher the frequency of action potentials produced.
Voltage recordings from an olfactory receptor cell during stimulation.
Odorants generate a slow receptor potential in the cilia; the receptor potential propagates along the dendrite and triggers a series of action potentials within the soma of the olfactory receptor cells. Finally, the action potentials (but not the receptor potential) propagate continuously along the olfactory nerve axon.
1.4. Five Modes of Sensory Detection
The source of the stimulus can be external or internal.
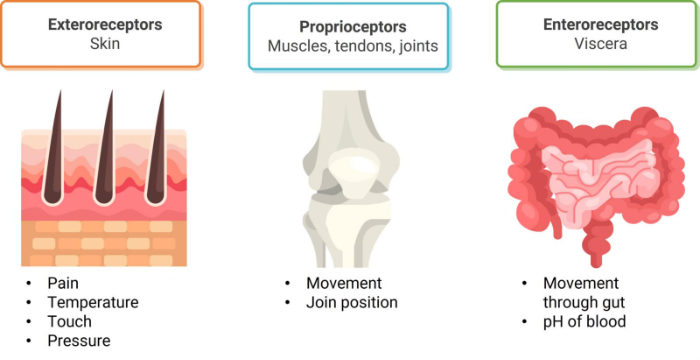
Superficially located sensory endings in the skin are called exteroceptors and respond to pain temperature, touch and pressure, that is stimuli outside the body.
Muscles, tendons, and joints have proprioceptors that signal awareness of body position and movement.
Enteroreceptors monitor events within the body such as feeling movement through the gut.
Mode of detection can be grouped into five categories:



1.5. Six Sensory Systems
Receptors are sensitive to particular forms of energy and receptor cells convert stimulus into electrical signals.
Neurons of the brain and spinal cord do not respond when they are touched or when they are exposed to sound or light or odors. Each form of energy must be transduced by a population of specialised cells, which converts the stimulus into a signal that all neurons understand.
In every sensory system, cells that perform this transduction step are called receptors. For each of the fundamental types of stimuli (mechanical, chemical, or thermal energy or light), there is a separate population of receptors selective for the particular form of energy.
Even within a single sensory system, there are class of receptors that are particularly sensitive to one stimulus (e.g., heat or cold) and not another (muscle stretch). The specificity in the receptor response is a direct function of differences in receptor structure and chemistry.
1.6. Stimulus Intensity
Sensory neurons use action potential frequency and duration to code stimulus intensity and duration.
Video 3: Stimulus Intensity
The intensity of a stimulus can not be directly calculated from a single sensory neuron action potential.
Watch the video below for more information.

Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.6
The intensity of a stimulus can not be directly calculated from a single sensory neuron action potential because a single action potential is “all-or-none”. Instead, stimulus intensity is coded in two types of information: the number of receptors activated and the frequency of action potentials coming from those receptors (frequency coding).
For individual sensory neurons, intensity discrimination begins at the receptor. If a stimulus is below threshold, the primary sensory neuron does not respond. Once stimulus intensity exceeds threshold, the primary sensory neuron begins to fire action potentials. As stimulus intensity increases, the receptor potential amplitude (strength) increase in proportion, and the the frequency of action potential in the primary sensory neurons increases, up to a maximum rate.
Similarly, the duration of a stimulus is coded by the duration of action potentials in the sensory neuron. In general, a longer stimulus generate s a longer series of action potential.
1.7. Two Modes of Receptor Adaptation
The two modes of receptor adaptation include:
- Tonic - slowly adapting
- Phasic - rapidly adapting
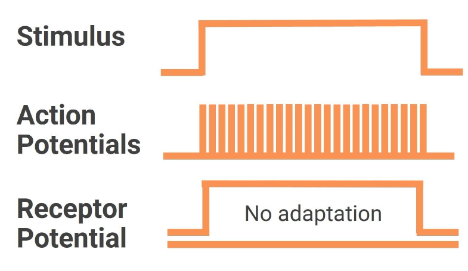
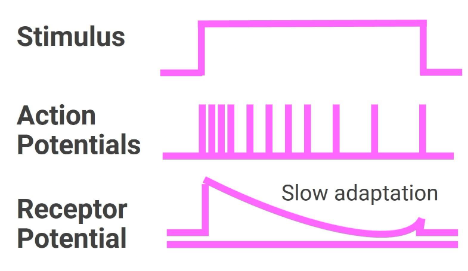
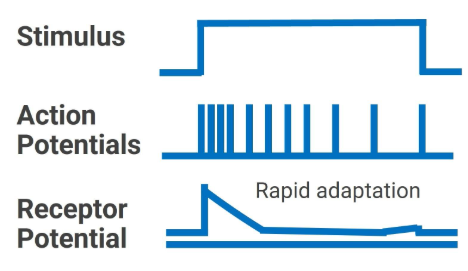
In general, the stimuli that activate tonic receptors are parameters that must be monitored continuously by the body. Once a stimulus reaches a steady intensity, phasic receptors adapt to a new steady state and turn off.
1.8. Receptor Response
Receptor response can decline with maintained stimuli: adaptation.
The rate of firing of this neuron, whose receptive field is located on the fifth finger, is rapid when the stimulus is first applied, but then adapts, slowing to a steady rate.
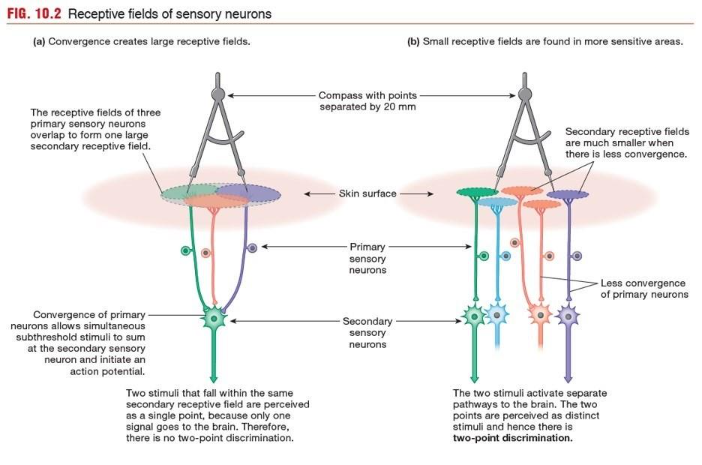
1.9. Receptive Fields of Sensory Neurons
A sensory neuron has a receptive field.
Somatic sensory and visual neurons are activated by stimuli that fall within a specific physical area known as the neuron’s receptive field.
The size of secondary receptive fields determines how sensitive a given area is to a stimulus.
1.10. Mach Band Effect
The Mach Band Effect is named after Ernst Mach.

Reference: https://en.wikipedia.org/wiki/Mach_bands
For each block of the color, are they the same or gradient, or? What do you think you see?
Lateral inhibition enhances contrast and makes a stimulus easier to perceive.
Video 4: Lateral Inhibition
Lateral inhibition, which increases the contrast between activated receptive fields and their inactive neighbors, is another way of isolating the location of a stimulus.
Watch the video below for more information.

Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.5
1.11. Labeled Lines Hypothesis
Each receptor (cold, warmth, touch, pain) has a distinct pathway linking the receptor surface to the brain, so different qualities of skin stimulation can be communicated to distinct places in the brain.
Video 5: Labeled Lines
The brain associates a signal coming form a specific group of receptors with a specific modality. This 1:1 association of a receptor with a sensation is called labeled line coding.
Watch the video below for more information.

One exception: the brain uses timing differences rather than specific neurons to localise sound.
Neurons in the ears are sensitive to different frequencies of sound, but they have no receptive fields and their activation provides no information about the location of the sound.
How does the brain figure out where the sound is from?

1.12. How does the CNS integrate sensory information?
These are the sensory pathways in the brain. Most pathways except the olfactory pathway pass through the thalamus on their way to the cerebral cortex.
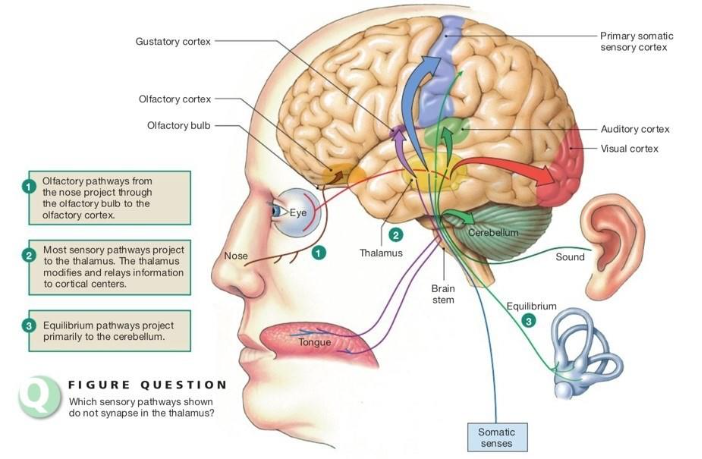
1.13. Summary: General Properties of Sensory Systems
1.14. Further Readings
-
- Bears et al., Neuroscience: exploring the brain, 4th edition, chapter 8 and 11
- Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, chapter 3
- Silverthorn, Human Physiology, 5th edition, chapter 10
Chapter 2: Special Senses (Taste, Smell and Hearing)
Introduction

How does the body perceive any smell, taste or sound?
How does the brain process and identify each of them?
Overview
In this chapter, we will look at the Special Senses, which include Chemoreception (Smell and Taste) and The Ear (Hearing).
Learning Objectives
At the end of this chapter, you should be able to:
-
1Describe the function of chemoreception and hearing2Identify the five taste sensations3Explain the function of olfactory receptor cells4Describe how sound is processed from the ear to the brain
2.1 What is Taste?
Taste is a combination of five basic sensations.
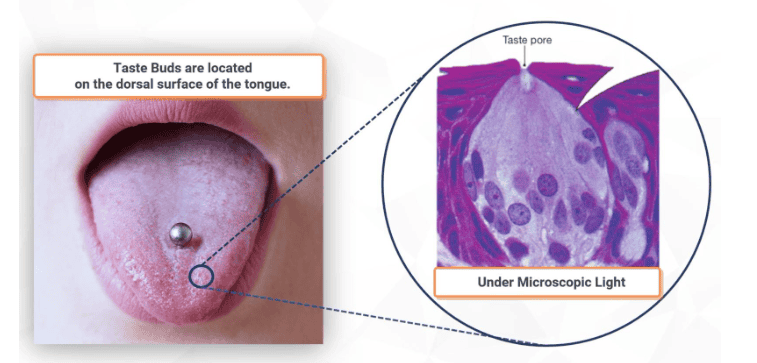
Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.14
Taste is a combination of five sensations: sweet, sour, salty, bitter and "umami", a taste associated with the amino acid glutamate and some nucleotides. "Umami" is a Japanese word which means deliciousness.
The receptors for taste are located primarily on taste buds clustered together on the surface of the tongue. One taste bud is composed of 50-150 taste cells.
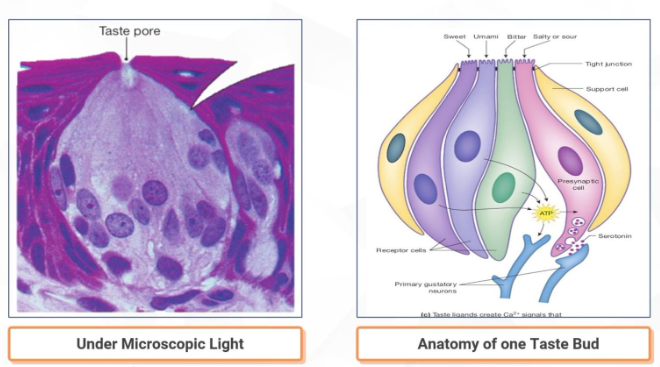
Each taste cell is a non-neural polarised epithelial cell tucked own into the epithelium so that only tiny tip protrudes into the oral cavity through a taste pore. In a given bud, tight junctions link the apical ends of adjacent cells together, limiting movement of molecules between the cells. The apical membrane of a taste cell is modified into microvilli to increase the amount of surface area in contact with the environment.
For a substance to be tasted, it must first dissolve in the saliva and mucus of the mouth. Dissolved taste ligands then interact with an apical membrane protein (receptor or channel) on a taste cell. Interaction of a taste ligand with a membrane protein initiates a signal transduction cascade that ends with a series of action potentials in primary neurons.
2.2. Mechanisms of Taste Transduction
The transduction mechanism for salt and sour tastants is different from the transduction mechanism for bitter, sweet and "umami" tastants.
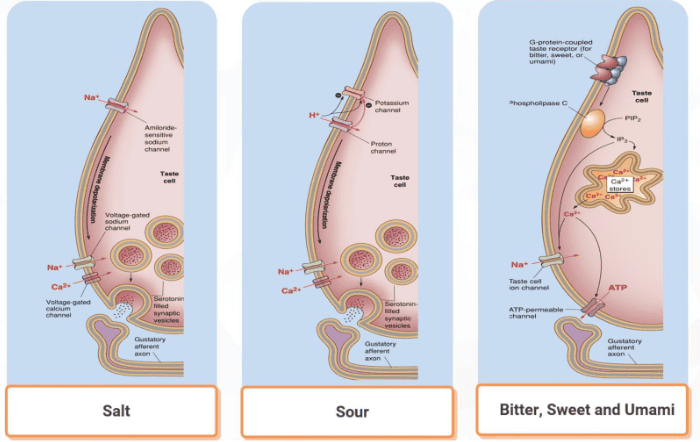
Transduction Mechanisms of Salt and Sour Tastants
Tastants can interact directly with ion channels either by passing through them (Na+ and H+) or by blocking them (H+ blocking the potassium channel). The membrane voltage then influences calcium channels on the basal membrane, which in turn influence the intracellular Ca2+ and transmitter release.
Transduction Mechanisms of Bitter, Sweet, and "Umami" Tastants
Tastants can interact directly with G-protein coupled membrane receptors, which activate phospholipase C, which increase the synthesis of IP3. IP3 then triggers the release of Ca2+ from internal storage sites, and Ca2+ opens a taste-specific ion channel, leading to depolarisation and transmitter release. The main transmitter is ATP, which is released from the taste cell by diffusing through ATP-permeable channels.
2.3. Oldest Senses - Olfaction
Video 6: Olfaction is One of the Oldest Senses
What defines an olfactory system? What does it do?
Watch the video below for more information.
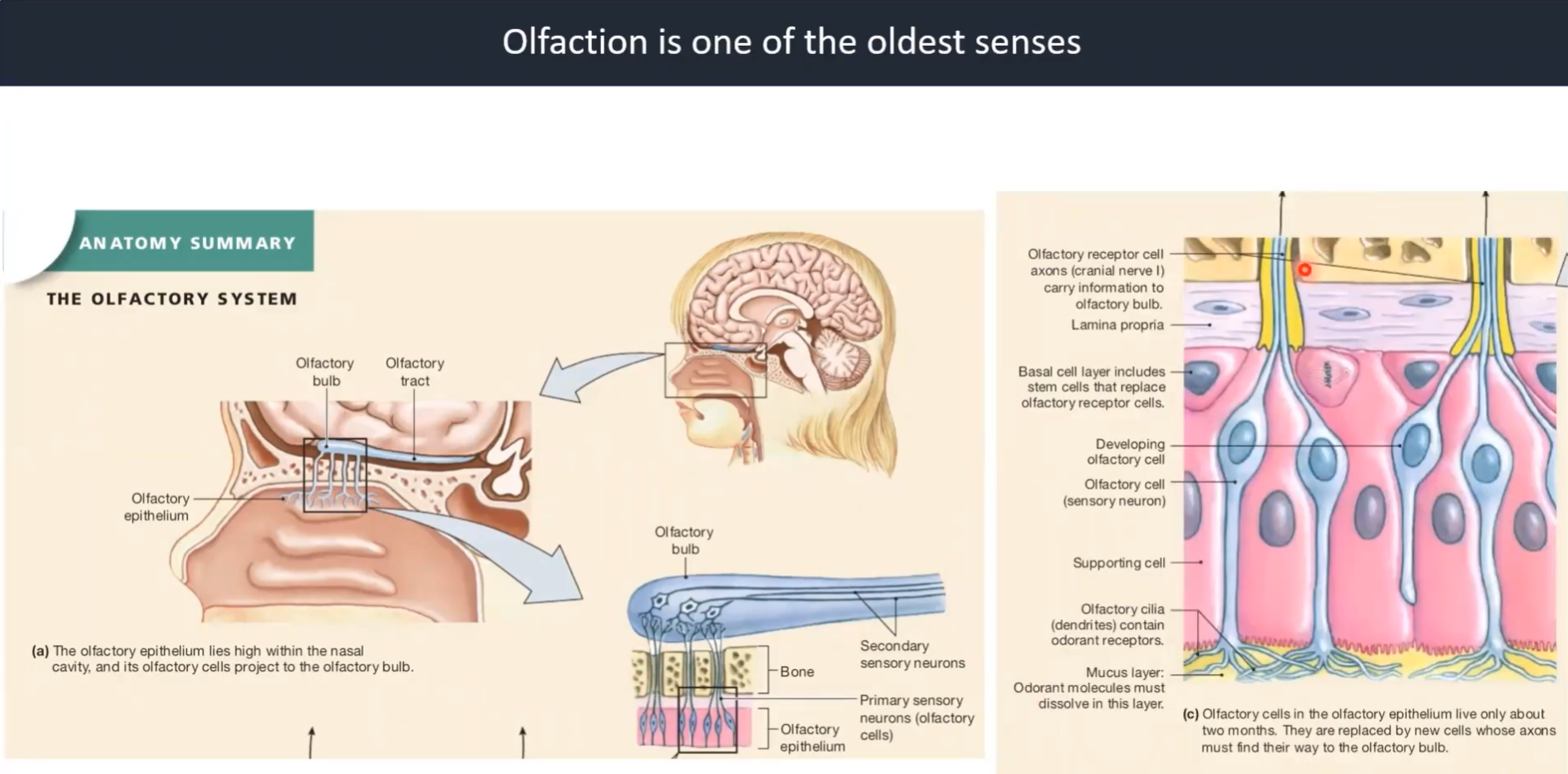
Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.13
2.4. Mechanism of Olfactory Transduction
Video 7: Mechanism of Olfactory Transduction
Have you wonder how the function of smell happen in human beings?
Watch the video below for more information.

Reference: Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 8.10
2.5. The Ear: Hearing and Equilibrium
The ear is a sense organ that is specialised for two distinct functions: hearing and equilibrium.
It can be divided into external, middle and inner sections. With the neurological elements housed in and protected by structures in the inner ear. The vestibular complex of the inner ear is the primary sensor for equilibrium. The reminder of the ear is used for hearing.

Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.15
The external ear consists of the outer ear, or pinna, and the ear canal. The pinna is another example of an important accessory structure to a sensory system, and it varies in shape and location from species to species, depending on the animals survival needs. The ear canal is sealed at its internal end by a thin membranous sheet of tissue called the tympanic membrane, or eardrum.
The tympanic membrane separates the external ear from the middle ear, an air-filled cavity that connects with the pharynx through the eustachian tube. The eustachian tube is normally collapsed, sealing off the middle ear, but it opens transiently to allow middle ear pressure to equilibrate with atmospheric pressure during chewing, swallowing, and yawn-ing. Colds or other infections that cause swelling can block the eustachian tube and result in fluid buildup in the middle ear. If bacteria are trapped in the middle ear fluid, the ear infection known as otitis media [oto-, ear * -itis, inflammation * media, middle] results.
Three small bones of the middle ear conduct sound from the external environment to the inner ear: the malleus [hammer], the incus [anvil], and the stapes [stirrup]. The three bones are connected to one another with the biological equivalent of hinges. One end of the malleus is attached to the tympanic membrane, and the stirrup end of the stapes is attached to a thin membrane that separates the middle ear from the inner ear.
The inner ear consists of two major sensory structures. The vestibular apparatus with its semicircular canals is the sensory transducer for our sense of equilibrium. The cochlea of the inner ear contains sensory receptors for hearing. On external view the cochlea is a membranous tube that lies coiled like a snail shell within a bony cavity. Two membranous disks, the oval window (to which the stapes is attached) and the round window, separate the liquid-filled cochlea from the air-filled middle ear. Branches of cranial nerve VIII, the vestibulocochlear nerve, lead from the inner ear to the brain.
2.6. Amplitude and Frequency in Sound Waves
Sound waves alternate peaks of compressed air and valleys where the air is less compressed.
Hearing is our perception of the energy carried by sound waves, which are pressure waves with alternating peaks of compressed air and valleys in which the air molecules are farther apart.
Sound is the brain’s interpretation of the frequency, amplitude, and duration of sound waves that reach our ears. Our brain translate frequency of sound waves (the number of wave peaks that pass a given point each second) into the pitch of a sound. Low-frequency waves are perceived as low-pitched sounds, such as the rumble of distant thunder. High-frequency waves create high-pitched sounds, such as the screech of finger-nails on a blackboard.
Sound waves are distinguished by their Amplitude (in dB) and Frequency (in Hz).
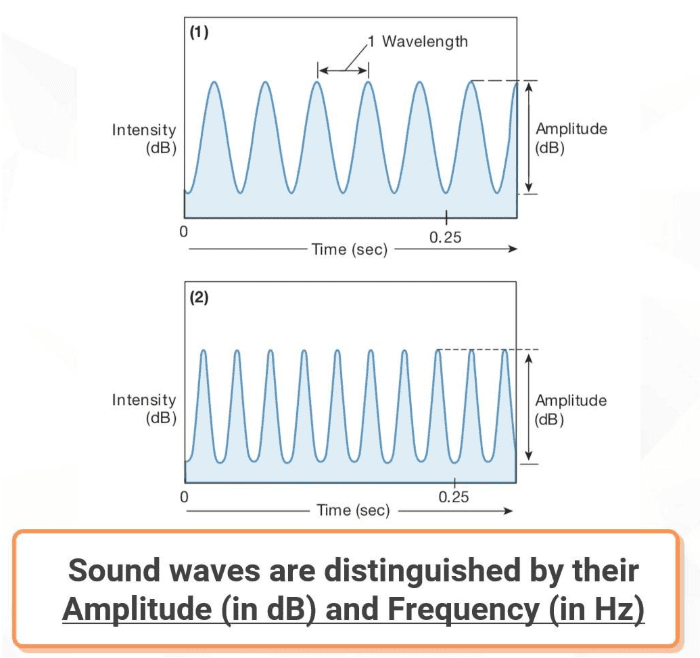
Sound wave frequency is measured in waves per second, or hertz (Hz). The average human ear can hear sounds over the frequency range of 20-20,000 Hz, with the most acute hearing between 1000-3000 Hz. Bats listen for ultra-high-frequency sound waves (in the kilohertz range) that bounce off objects in the dark. Elephants and some birds can hear sounds in the infrasound (very low frequency) range.
Loudness is our interpretation of sound intensity and is influenced by the sensitivity of an individual’s ear. The intensity of a sound wave is a function of the wave amplitude. Intensity is measured on a logarithmic scale in units called decibels (dB). Each 10-db increase represents a 10-fold increase in intensity.
Normal conversation has a typical noise level of about 60dB. Sounds of 80 dB or more can damage the sensitive hearing receptors of the ear, resulting in hearing loss.
2.7. Sound Transmission through the Ear
Video 8: Sound Transmission Through the Ear
Hearing is a complex sense that involves multiple transductions.
Watch the video below for more information.
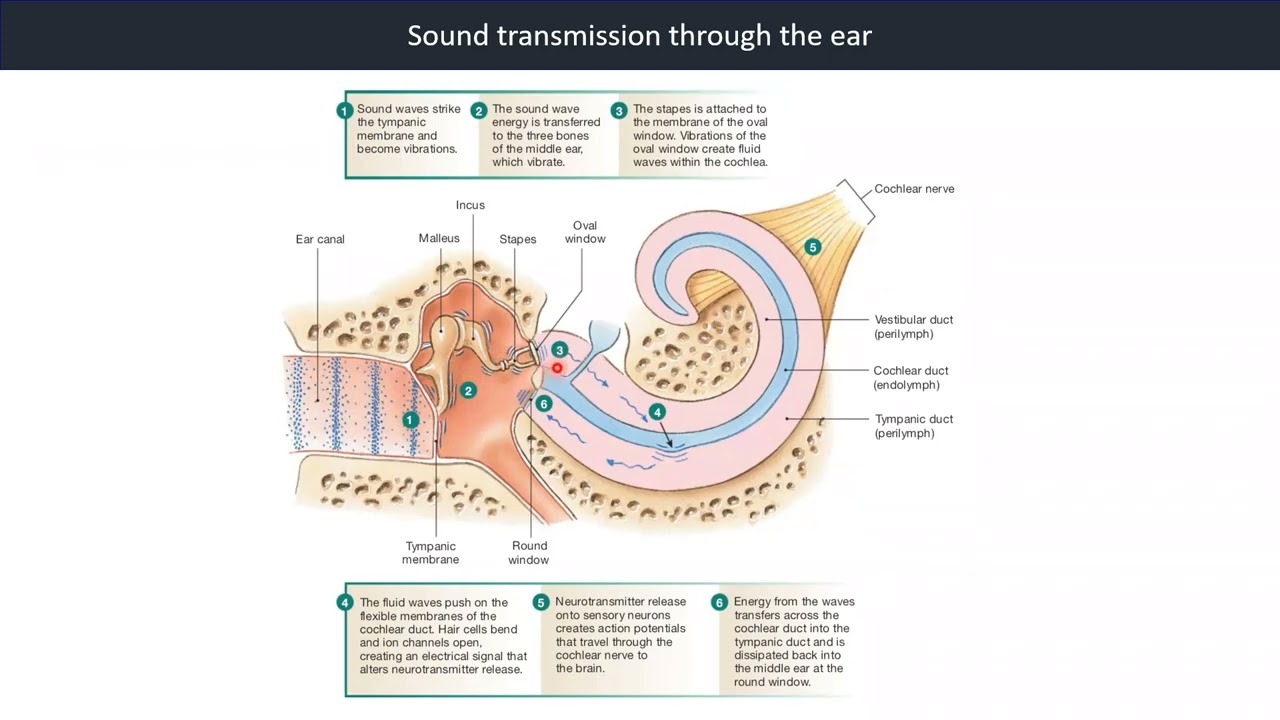
Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.17
2.8. Signal Transduction in Hair Cells
Video 9: Signal transduction in hair cells
Hair cells are non-neural receptor cells. The surface of each hair cell have about 50-100 stiffened cilia. The longest cilium is connected a tectorial membrane. If the tectorial membrane moves, the cilia does it too.
Watch the video below for more information.

Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.19
2.9. Sensory Coding for Pitch
The basilar membrane has variable sensitivity to sound wave frequency along the length.
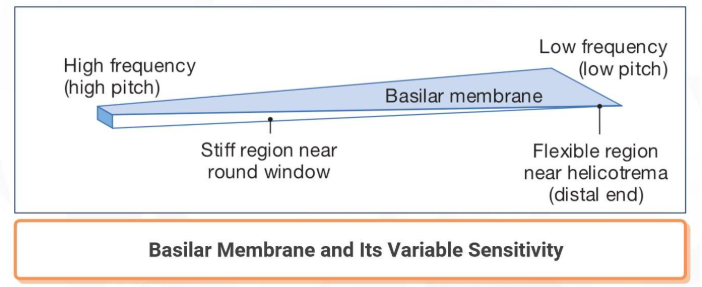
The auditory system processes sound waves so that they can be discriminated by location, pitch and loudness.
Initial processing for pitch and loudness takes place in the cochlea of each ear.
Coding sound for pitch is primarily a function of the basilar membrane. The membrane is tiff and narrow near its attachment between the round and oval windows but widens and becomes more flexible near its distal end.
The frequency of sound waves determines the displacement of the basilar membrane. The location of active hair cells creates a code that the brain translates as information about the pitch of sound.

High frequency waves entering the vestibular duct create maximum displacement of the basilar membrane close to the oval window and consequently are not transmitted very far along the cochlea. Low-frequency wave travel along the length of the basilar membrane and create their maximum displacement near the flexible distal end. The differential response to frequency transforms the temporal aspect of frequency (numbers of sound waves per second) into spatial coding for pitch by location along the basilar membrane.
A good analogy is a piano keyboard, where the location of a key tells you its pitch. The spatial coding of the basilar membrane is preserved in the auditory cortex as neurons project from hair cells to corresponding regions in the brain.
Loudness is coded by the ear in the same way the signal strength is coded in somatic receptors.
2.10. Summary: Special Senses
2.11. Further Readings
-
- Bears et al., Neuroscience: exploring the brain, 4th edition, chapter 8, 11 and 12
- Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, chapter 22
- Silverthorn, Human Physiology, 5th edition, chapter 10
Chapter 3: Somatosensory System - Touch
Introduction

Have you wondered how every object you touch feels different?
Why do you react in a certain when you touch cold and hot object?
Overview
Learning Objectives
3.1. Primary Afferent Axons, Spinal Cord and General Projection Pathway
How does your body know how and when to react to a touch stimuli?
Peripheral Nervous System (PNS)
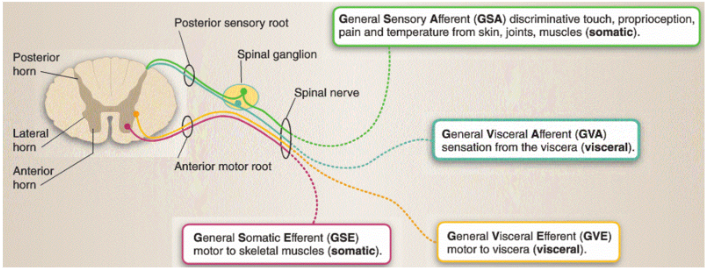
Sensory information enters the spinal cord through the posterior roots. Sensory cell bodies lie in the spinal ganglion of each spinal nerve. Motor information leaves the spinal cord through the anterior roots, and LMNs are located in the anterior horn at each spinal level. Each segment of the spinal cord innervates a specific area of skin, referred to as a dermatome, and a specific muscle group, referred to as a myotome. Primary efferent autonomic fibers have their cell bodies in the lateral horn and leave the spinal cord through the anterior root.The afferent autonomic fibers travel with the somatic afferents through the posterior root.
Pathway for Somatic Perception
Video 10: Somatic Perception to Somatosensory Cortex and Cerebellum
Sensory systems bring the information to an individual. The pathways for somatic perception projects to the somatosensory cortex and cerebellum.
Watch the video below for more information.

Reference: Silverthorn, Human Physiology, 5th edition, Chapter 10, Figure 10.8 and 10.9
3.2. Mechanoreceptors of the Skin
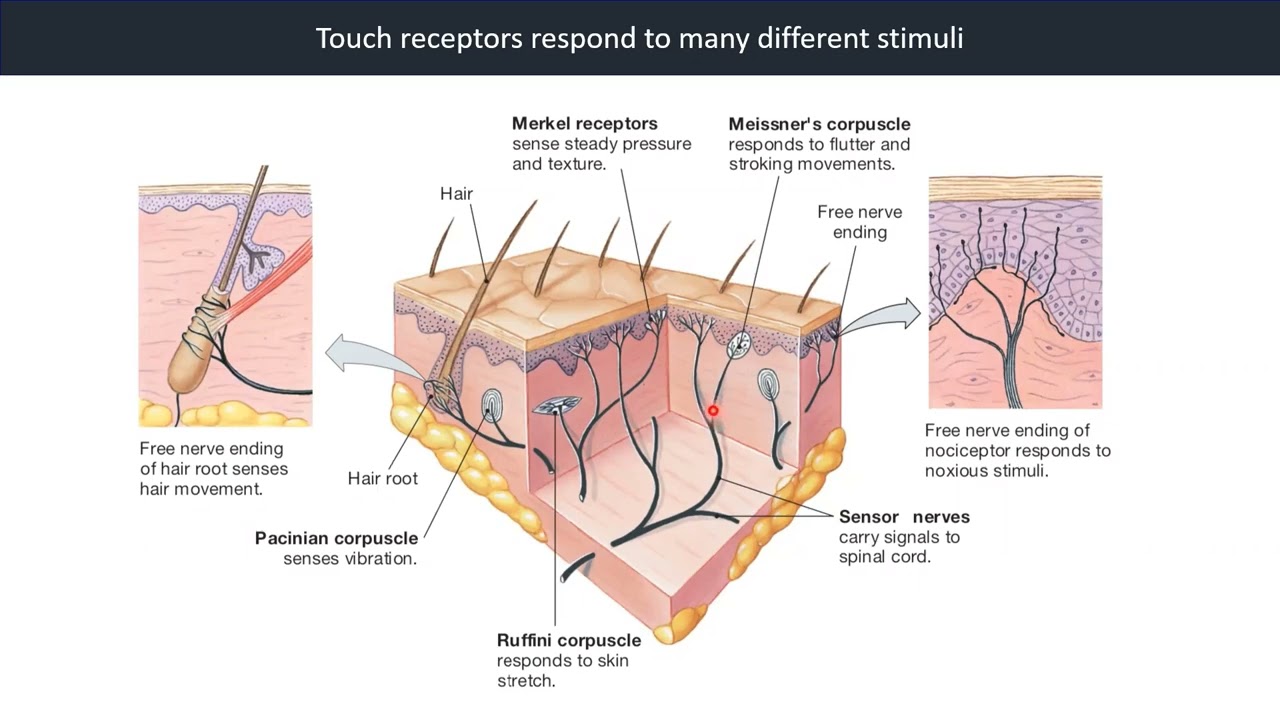
Touch receptors respond to many different stimuli.
Video 11: Touch Receptors in the Skin
The receptors are distributed throughout the body. Of all, the touch receptors are among the most common receptors in the body.
With the various forms of touch receptors, watch the video below for more information.
Receptive Fields of Human Sensory Receptors Test
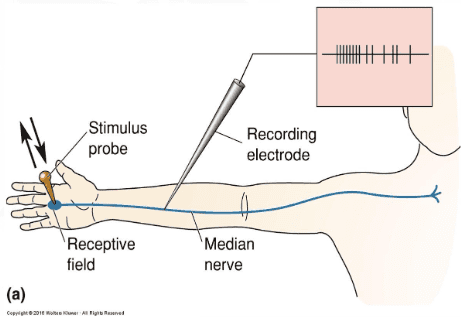
By introducing a microelectrode into the media nerve of the arm, it is possible to record the action potentials from a single sensory axon and map its receptive field on the hand with a fine stimulus probe.
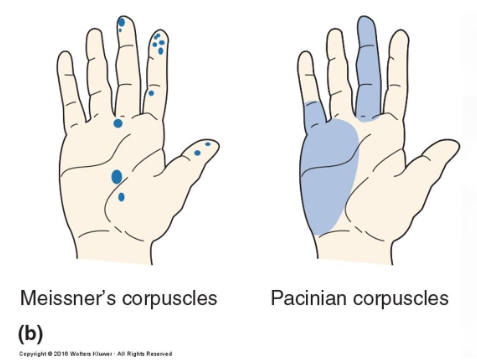
Results show that receptive fields are either relatively small as in Messner’s corpuscles, or large, as in Pacinian Corpuscle.
Sensory Receptors
Video 12: Sensory Receptors in the Skin
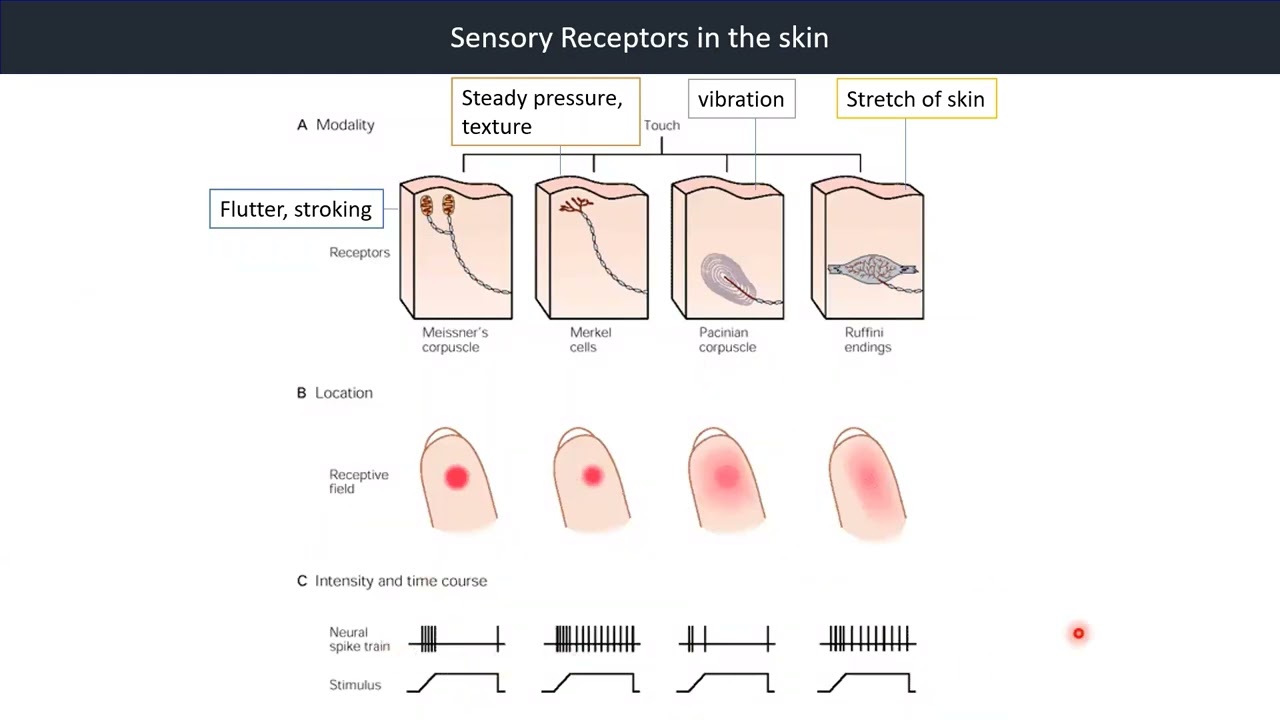
Sensory receptors exist in all layers of the skin. There are four primary mechanoreceptors in human skin: Meissner's corpuscles, Merkel cells, Pacinian corpuscle and Ruffini endings.
Watch the video below for more information.
Mechanosensitive Ion Channels
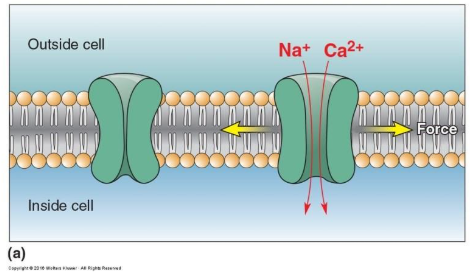
Some membrane ion channels are sensitive to stretching of the lipid membrane, tension in the membrane directly induced the channel to open and allow cations to flow.
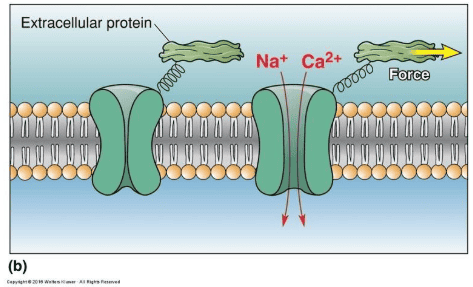
Other ion channels open when force is applied to extracellular structures linked to the channels by peptides.
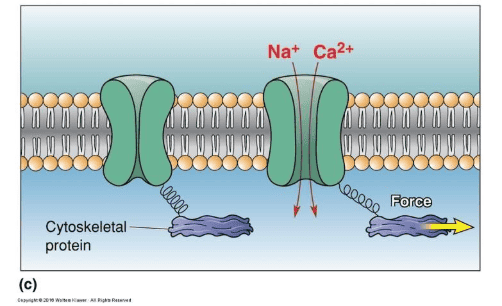
Mechanically sensitive channels may also be linked to intracellular proteins, especially those of the cytoskeleton; deformation of the cell and stress on its cytoskeleton generate forces that regulate channel gating.
3.3. Thermoreceptors
Temperature Receptors as Free Nerve Endings

Temperature receptors are free nerve endings that terminate in the subcutaneous layers of the skin. Cold receptors are sensitive primarily to temperatures lower than body temperature. Warm receptors are stimulated by temperatures in the range extending from normal body temperature (37° C) to about 45° C. Above that temperature, pain receptors are activated, creating a sensation of painful heat.
Thermoreceptor Transient Receptor Potential (TRP) Channels
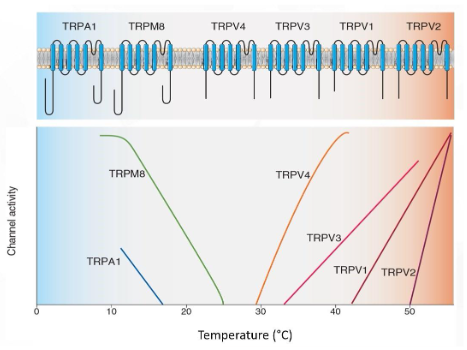
Thermoreceptor TRP channels tuned to detect different temperatures.
Reference: Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.35
The arrangement of the known thermosensitive TRP channel protein molecules in the neuronal membrane. TRPM8 and TRPV1 are responsive to menthol and capsaicin, respectively.
The graph plots the activation of the various TRP channels as a function of temperature.
Adaptations of Thermoreceptors

The receptive field for a thermoreceptor is about 1 mm in diameter, and the receptors are scattered across the body. There are considerably more cold receptors than warm ones. Temperature receptors slowly adapt between 20° and 40° C. Their initial response tells us that the temperature is changing, and their sustained response tells us about the ambient temperature.
Outside the 20-40° C range, where the likelihood of tissue damage is greater, the receptors do not adapt.
Adaptations of thermoreceptors. The responses of cold and warm receptors to a step reduction in skin temperature are shown. Both receptors are most responsive to sudden changes in temperature, but they adapt over several seconds.
Cold receptors are coupled to A-delta and C fibers, while warm receptors are coupled only to C fibers.
3.4. Anatomical Pathways
Structure of A Segment of The Spinal Cord and Its Roots

The spinal cord is made of gray and white matter. The gray matter, located in the central part of the spinal cord, is shaped like a butterfly. The white matter surrounds the gray matter and is made of axons. These axons contains pathways that connects the brain with the other parts of the body.
The pathway in red shows the path taken by the primary afferent axons. We will take a closer look at the primary afferent axons in the next segment.
Primary Afferent Axons
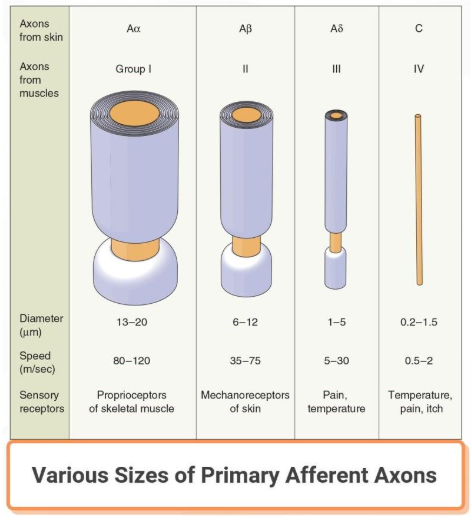
The primary afferent axons are nerve fibers connected to different types of receptors around the body. There are various sizes of primary afferent axons. They are drawn to scale, but they are shown 2000 times larger than life size. The diameter of an axon is correlated with its conduction velocity and with the type of sensory receptor to which it is connected.
The different nerve fiber groups are A-alpha, A-beta, A-delta and C-nerve fibers.
A-alpha, A-beta and A-delta nerve fibers are insulated with myelin. C-nerve fibers are unmyelinated. It is believed the thicker a nerve fiber the faster information travels. Vice versa, the thinner a nerve fiber, the slower information travels.
Segmental and Longitudinal Organisation
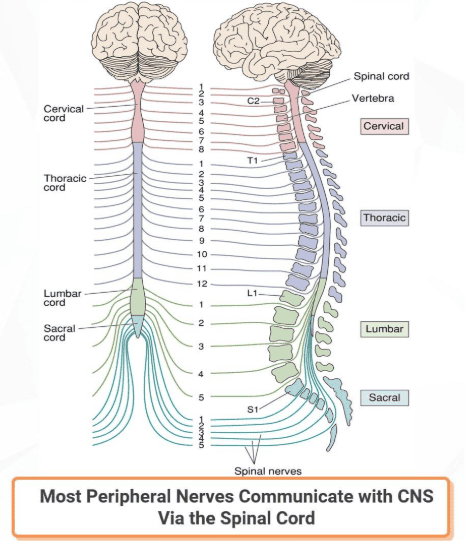
The spinal cord is divided into four different regions: the cervical, thoracic, lumbar and sacral regions. Each spinal nerve, consisting of dorsal root and ventral root axons, passes through a notch between the vertebrae (the "back bones") of the spinal column.
The 30 spinal segments are divided into 4 groups. Each segment is named after the vertebra adjacent to where the nerves originate: cervical (C) 1-8, thoracic (T) 1-12, lumbar (L) 1-5, and sacral (S) 1-5.
Dorsal and ventral roots enter and leave the vertebral column through intervertebral foramen at the vertebral segments corresponding to the spinal segment. The arrangement of paired dorsal and ventral roots is repeated 30 times down the length of the human spinal cord.
Dermatomes
Video 13: Dermatomes
A dermatome is the area of skin triggered by right and left dorsal roots of a spinal segment. If a nerve is cut, one loses sensation from that dermatome.
Watch the video below for more information.

Reference: Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.11
The Dorsal Column-Medical Leminiscal Pathway
Video 14: The Dorsal Column-Medical Leminiscal Pathway
The dorsal column-medial lemniscal pathway is the major route by which touch and proprioceptive information ascend to the cerebral cortex.
Watch the video below for more information.

Reference: (1) Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.15; (2) Krebs et al., Lippincott’s Illustrated
Reviews: Neuroscience, Figure 13.9; (3) Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.18
3.5. Somatosensory Cortex and Plasticity
Somatosensory Cortex
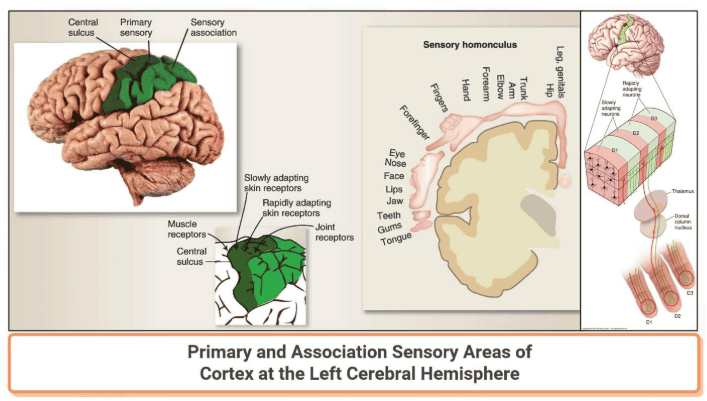
The somatosensory cortex is the part of the brain that recognizes where ascending sensory tracts originate. Each sensory tract has a corresponding region of the cortex, so that all sensory pathways for the left hand terminate in one area, all pathways for the left foot terminate in another area, and so on. Within the cortical region for a particular body part, columns of neurons are devoted to particular types of receptors. For example, a cortical column activated by cold receptors in the left hand may be found next to a column activated by pressure receptors in the skin of the left hand. This columnar arrangement creates a highly organized structure that maintains the association between specific receptors and the sensory modality they transmit.
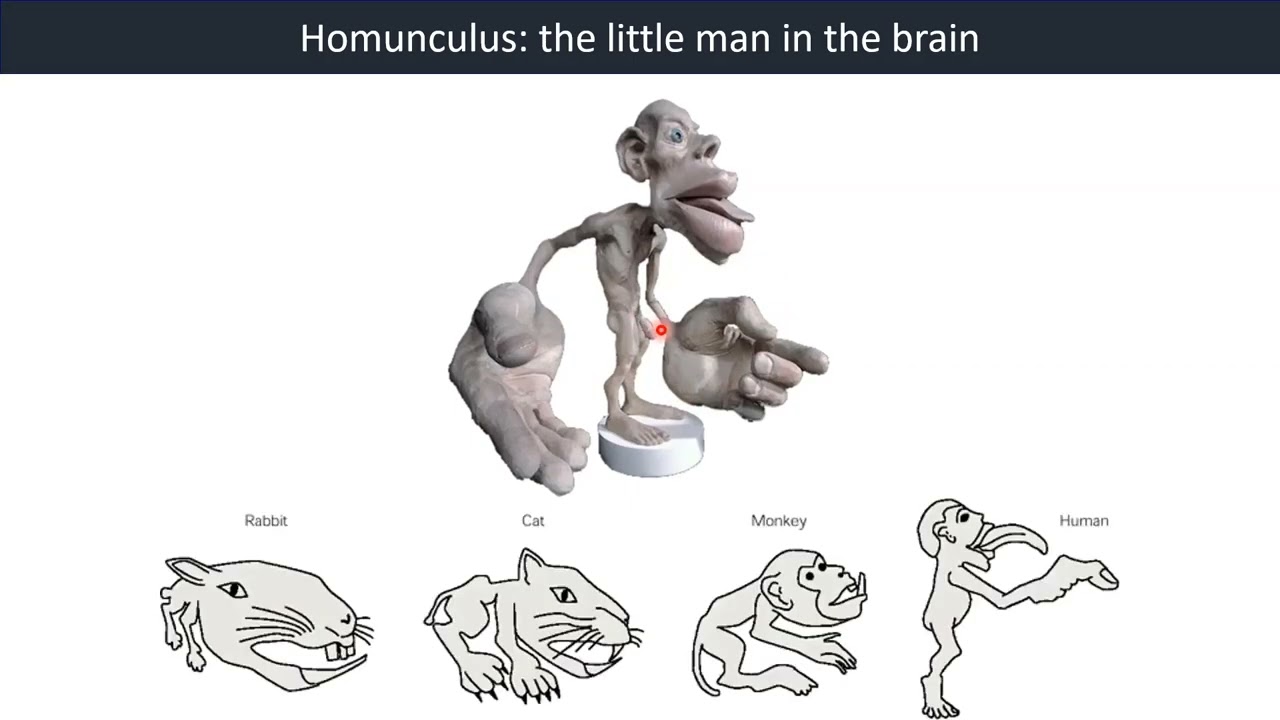
Homunculus
Video 15: Homunculus - The Little man in the Brain
Watch the video below for more information.
Somatotopic Map Plasticity
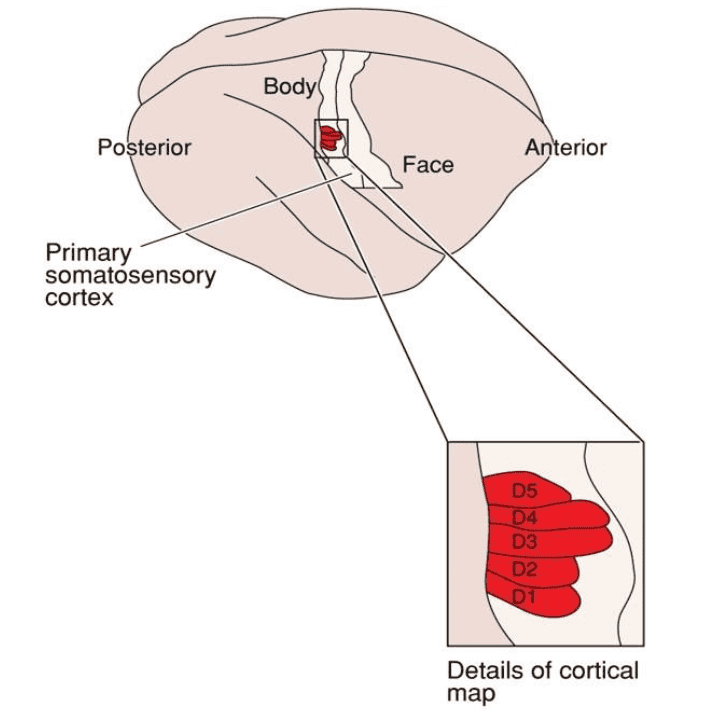
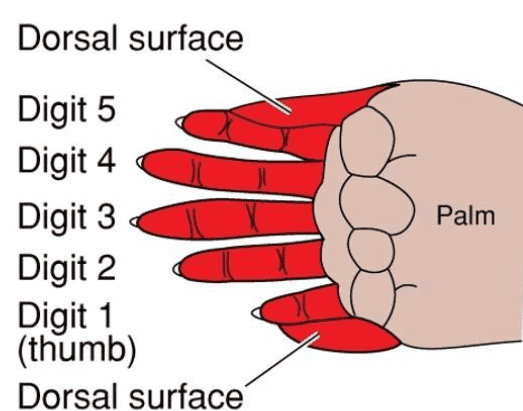

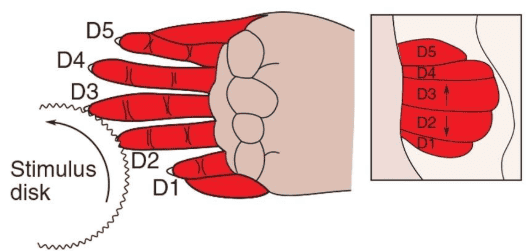
D. Reorganisation of Cortical Map after Discrimination Training of Two Fingertips
Reference: Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.23
Does reorganisation of afferent fibers also occur in the human brain?
Magnetoencephalography can now be used to construct functional maps of the hand in normal subjects with a precision of millimeters. This imaging technique has been used to compare the hand area in the cortex of normal adult humans to that of patients with a congenital fusion of the fingers (syndactyly). Patients with this syndrome do not have individual fingers—their hand is much like a fist—so that neural activity in one part of the hand is always correlated with activity in all other parts of the hand. The size of the representation in the cortex of the syndactylic hand is considerably less than that of a normal person, and within this shrunken representation the fingers are not organized somatotopically, as are separate fingers.
In contrast, if a person loses a finger or limb, the portion of the somatosensory cortex devoted to the missing structure begins to be taken over by sensory fields of adjacent structures. Reorganization of the somatosensory cortex map is an example of the remarkable plasticity of the brain. Unfortunately, sometimes the reorganization is not perfect and can result in sensory sensations, including pain, that the brain interprets as being located in the missing limb (phantom limb pain).
Phantom Limb Sensations
Phantom limb sensations can be evoked by touching the face. A common experience among amputees is the perception of sensations coming from the missing limb when other body parts are touched.
-
- A subject whose arm was amputated above the left elbow shows sites on his face where stimulation (brushing the face with a cotton swab) elicits sensation referred to the phantom digits. Regions of the body that evoke referred sensations are called reference fields. Stimulation of the region labeled T always evoked sensations in the phantom thumb. Stimulation of facial areas marked I, P, and B evoked sensation in the phantom index finger, pinkie, and ball of the thumb, respectively. This patient was tested four weeks after amputation.
- The upper arm of a subject who experienced referred sensation in the face and in two distinct areas on the arm—one area close to the line of amputation and a second area 6 cm above the elbow crease. Each area is a precise spatial map of the lost digits; the maps are almost identical except for the absence of fingertips in the upper map. When the patient imagined pronating his phantom lower arm, the entire upper map shifted in the same direction by about 1.5 cm. Stimulating the skin region between these two maps did not elicit sensations in the phantom limb.
- Portion of sensory homunculus showing how the cortical area receiving inputs from the hand is flanked by the regions devoted to the face and the arm. Rearrangement of these cortical inputs is thought to be responsible for some types of phantom limb sensation.
3.6. Summary: Somatosensory System - Touch
3.7. Further Readings
-
- Bears et al., Neuroscience: exploring the brain, 4th edition, chapter 8, 11 and 12
- Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, chapter 22
- Silverthorn, Human Physiology, 5th edition, chapter 10
Chapter 4: Physiological and Patho-Physiological Basis of Pain
Introduction

Have you felt "in pain"?
How does the brain process this sensation?
Was there an instance when your pain was alleviated by something?
How do analgesics work?
Overview
4.1. Nociceptors and the Transduction of Painful Stimuli
Nociceptors initiate protective responses
Nociceptors, which came from the word "nocere", "to injure", are receptors that respond to a variety of strong noxious stimuli (chemical, mechanical, or thermal) that cause or have the potential to cause tissue damage.



Nociceptors are sometimes called pain receptors, even though pain is a perceived sensation rather than a stimulus.
Nociceptive pain is mediated by free nerve endings whose ion channels are sensitive to a variety of chemical, mechanical, and thermal stimuli. For example, the membrane channels called vanilloid receptors (also called transient receptor potential V1 or TRPV1 channels) respond to damaging heat from a stove or other source, as well as to capsaicin, the chemical that makes hot chili peppers burn your mouth.
At the opposite end of the temperature spectrum, researchers recently identified a membrane protein that responds both to cold and to menthol, one reason mint-flavored foods feel cool. Pain can be felt in skeletal muscles (deep somatic pain) as well as in the skin.
Muscle pain during exercise is associated with the onset of anaerobic metabolism and is often perceived as a burning sensation in the muscle (as in go for the burn!). Some investigators have suggested that the exercise-induced metabolite responsible for the burning sensation is K*, known to enhance the pain response. Muscle pain from ischemia (lack of adequate blood ow that reduces oxygen supply) also occurs during myocardial infarction (heart attack).
Video 16: Nociceptive Fibers
Nociceptors can be activated by extreme temperature, intense mechanical stimulation, or an array of chemicals through specific receptors.
Watch the video below for more information.
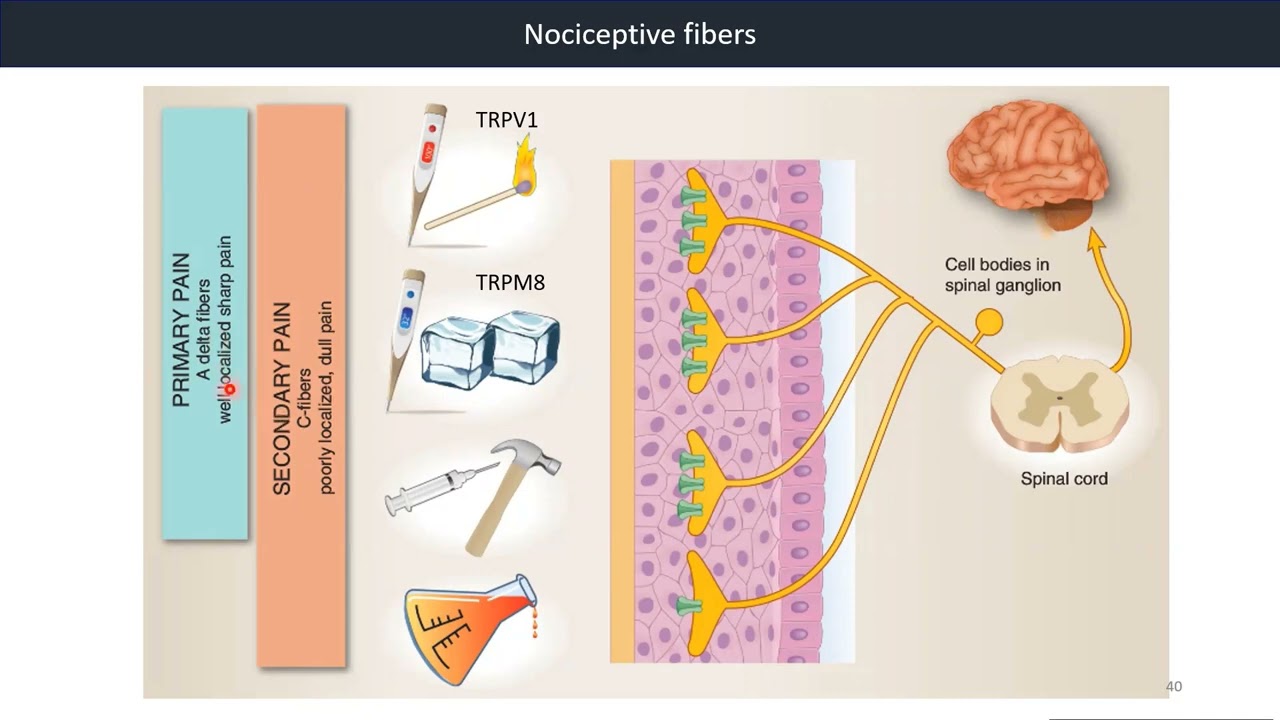
Reference: (1) Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, Figure 22.1; (2) Bears et al., Neuroscience: exploring the brain, 4th edition,
Figure 12.25; (3) Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.27
4.2. Referred Pain: Viscerosomatic Convergence
Nociceptor axons from the viscera enter the spinal cord by the same route as the cutaneous nociceptors.
Within the spinal cord, there is a substantial mixing of information from these two sources of input. This cross-talk gives rise to the phenomenon of referred pain, where visceral nociceptor activation is perceived as a cutaneous sensation.

Pain in the heart and other internal organs (visceral pain) is often poorly localized and may be felt in areas far removed from the site of the stimulus.
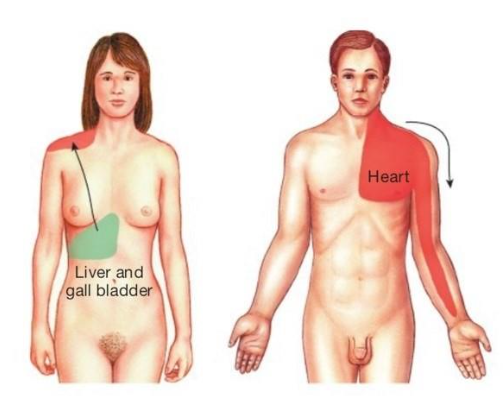
For example, the pain of cardiac ischemia may be felt in the neck and down the left shoulder and arm. This referred pain apparently occurs because multiple primary sensory neurons converge on a single ascending tract.
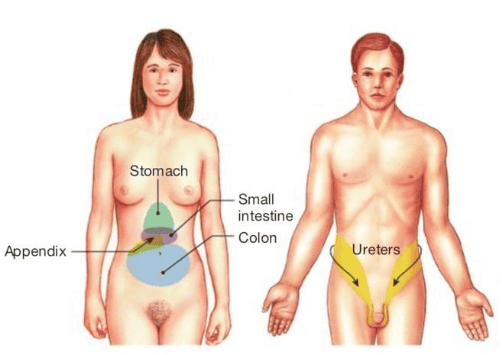
According to this model, when painful stimuli arise in visceral receptors, the brain is unable to distinguish visceral signals from the more common signals arising from somatic receptors. As a result, it interprets the pain as coming from the somatic regions rather than the viscera.
4.3. Sensitisation of Peripheral Receptors
Tissue damage and inflammation result in the release of inflammatory molecules, such as bradykinin and prostaglandins, which sensitise peripheral nociceptors.
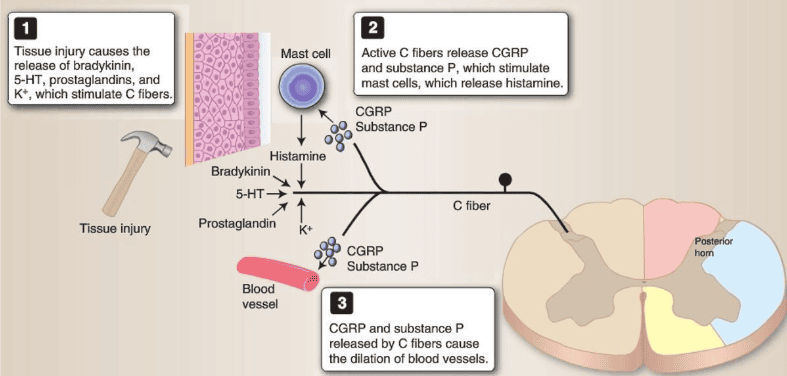
In addition, when a noxious stimulus is detected by a nociceptor, free nerve endings will release substance P and CGRP. These two neuropeptides contribute to the inflammatory response at the site of tissue injury by stimulating mast cell release of histamine and bradykinin. CGRP induces vasodilation, which results in further release of inflammatory molecules.
Nociceptor activation is modulated by local chemicals that are released upon tissue injury, including K*, histamine, and prostaglandins released from damaged cells; serotonin released from platelets activated by tissue damage; and the peptide substance P, which is secreted by primary sensory neurons.
These chemicals, which also mediate the inflammatory response at the site of injury, either activate nociceptors or sensitize them by lowering their activation threshold. Increased sensitivity to pain at sites of tissue damage is called in inflammatory pain.
Nociceptors may activate two pathways
- Reflexive protective responses that are integrated at the level of the spinal cord
- Ascending pathways to the cerebral cortex that become conscious sensation (pain or itch)
Primary sensory neurons from nociceptors terminate in the dorsal horn of the spinal cord. There they synapse onto secondary sensory neurons that project to the brain or onto interneurons for local circuits.
4.4. Projection of Fibers into the Spinal Cord
The synaptic targets of Ad and C fibers are either nociceptive-specific (NS) cells, which synapse only with Ad and C fibers, or wide dynamic range (WDR) neurons, which receive synaptic input from all types of sensory fibers.

Whereas NS neurons will encode only for painful stimuli and project to higher centers, the WDR cells can encode for a range of stimuli, both painful and non-painful.
Neurotransmitter
The synapse in the posterior horn is excitatory. The neurotrasmitters released by the afferent nociceptive fibers are glutamate, which acts mainly on a-amino-3-hydroxy-5-methyl-4-isoxazaole propionic acid (AMPA) and N-methyl-d-aspartic acid (NMDA) receptors; substance P, which acts on the NK1 receptor, and GRP, which also has an excitatory effect via the CGRP receptor.
Wide Dynamic Range Neurons
WDR cells can fire Aps in a graded fashion, depending on the stimulus intensity. Stimulus intensity is encoded by the frequency of C-fiber signaling: the more painful the stimulus, the higher the frequency of C-fiber discharge and the greater the WDR neuron response. The WDR neuron can then amplify this signal through a mechanism called “wind-up”.
Video 17: Projection of Fibers into the Spinal Cord
Watch the video below for more information.
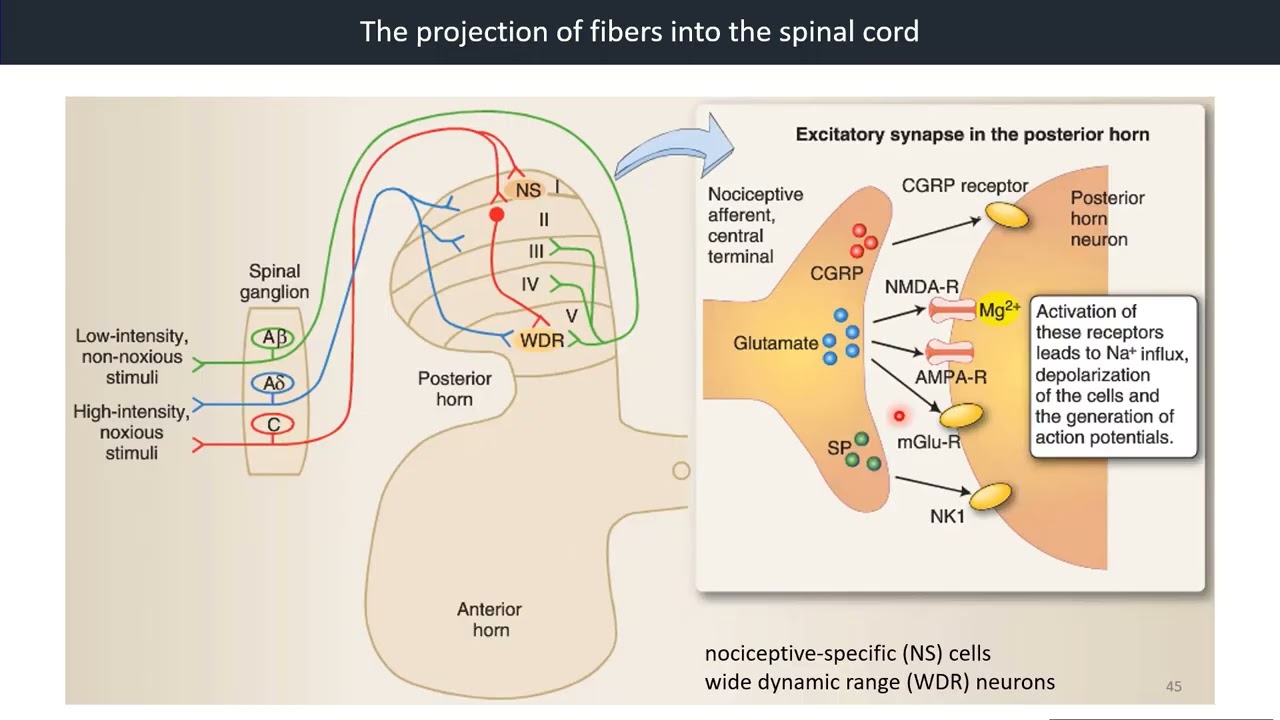
Reference: Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, Figure 22.3
4.5. Principles of Central Sensitisation “Wind-up”
Wind-up: Central sensitization, or “wind-up,” in the spinal cord is mediated through neurotransmitter release at the postsynaptic neuron in the posterior horn.
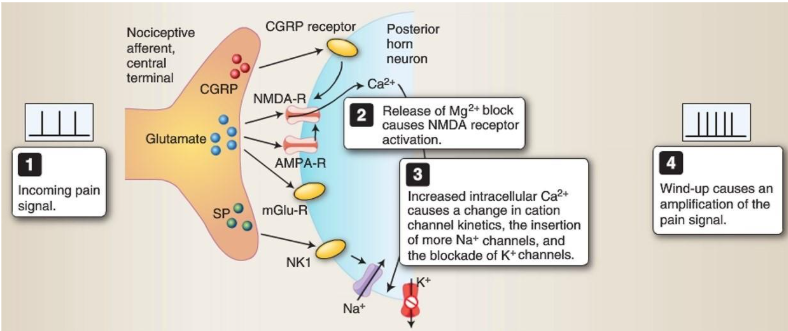
Wind-up:
- is essentially an amplification system within the spinal cord to respond to the cumulative nociceptive input from C fibers.
- results from the repetitive excitatory stimulation of the WDR neuron through glutamate acting mainly on AMPA receptors.
This increased AP frequency and sustained membrane depolarization result in activation of NMDA receptors. The NMDA receptor is usually inactive due to blockade of channels by Mg2+ ions. Sustained depolarization releases this Mg2+ block, and the NMDA receptor can then be activated by glutamate. Significantly, because the NMDA receptor is Ca2+ permeable, Ca2+ influx into the cell changes the electrophysiological signaling properties of the WDR neuron.
4.6. Hyperalgesia and Allodynia
When the response to a normally painful stimulus is heightened (more pain felt than the usual).
When a normally nonpainful stimulus is perceived as painful (for example, light touch on sunburn).
4.7. Ascending Pain Pathways
Nociceptors may activate two pathways
Reflexive protective responses that are integrated at the level of the spinal cord, with ascending pathways to the cerebral cortex that become conscious sensation (pain or itch).
Video 18: Ascending Pain Pathways
Watch the video below for more information.
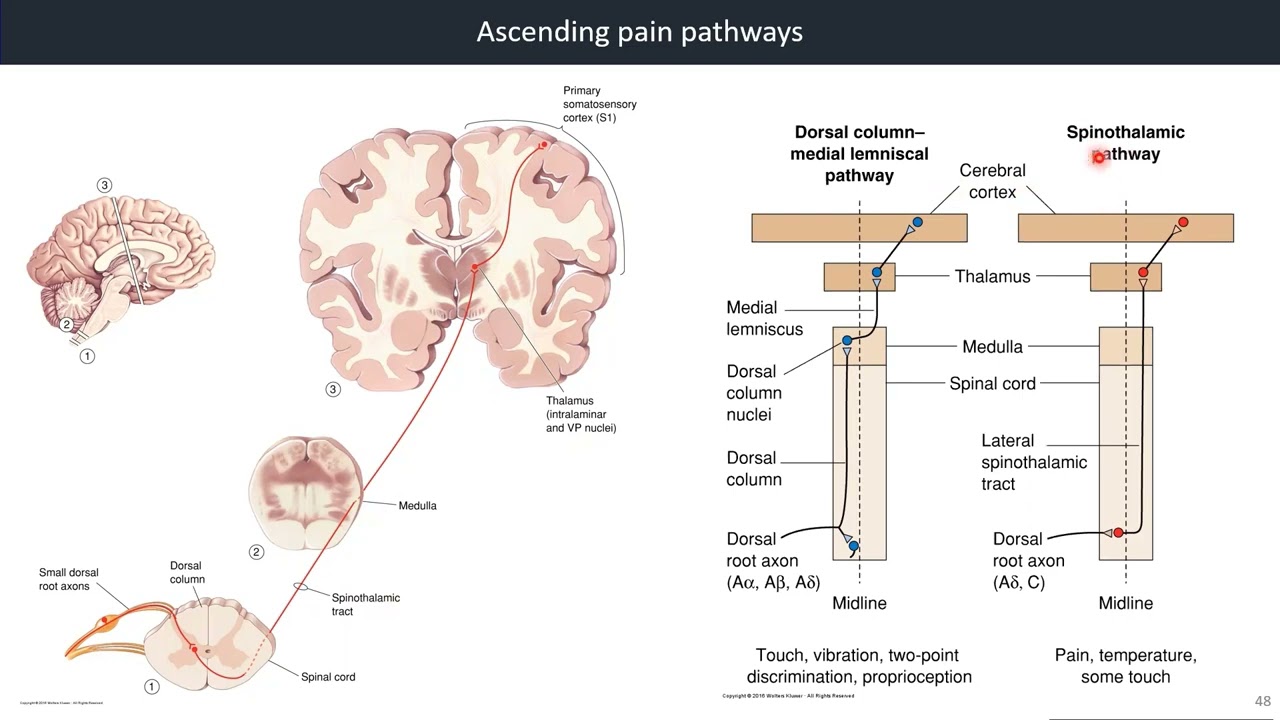
Reference: Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.30 & 12.31
4.8. Medial Affective-Motivational Pathway vs Lateral Sensory-Discriminative Pathway
Medial Affective-Motivational Pathways
- These pathways influence the emotional and visceral responses to pain as well as the descending modulation of pain.
- The medial affective-motivational component of the system is the “second” pain, the dull, throbbing poorly localised type of pain.
- This component includes the emotional response to pain: “Ouch!! That hurts—I don’t like it!”
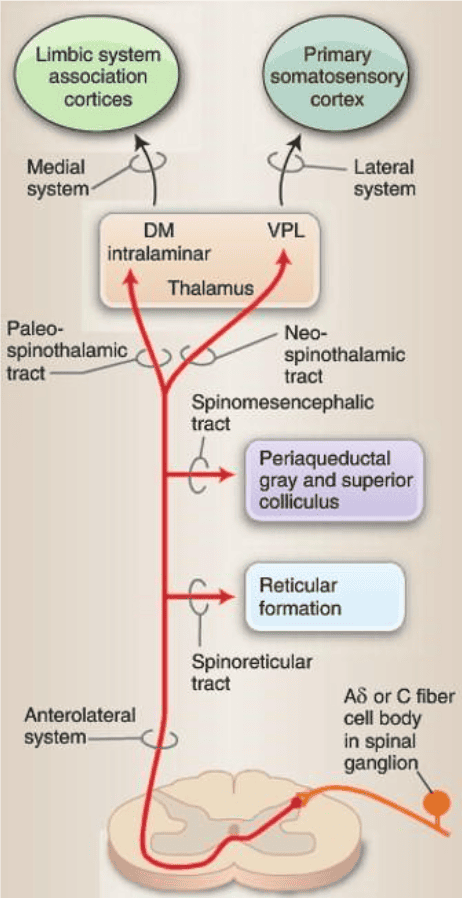
Lateral Sensory-Discriminative Pathway
- The tract is organized somatotopically and allows for the localization of pain in the primary somatosensory cortex.
- The lateral sensory-discriminative pathway mediates the “first” pain, the sharp, well-localized sensation relayed rapidly to the cortex.
- “I have a sharp pain in my left arm!”
Video 19: Medial System vs Lateral System
The ascending pathways for nociception cross the body's midline in the spinal cord and ascend to the thalamus and sensory areas of the cortex. The pathways also send branches to the limbic system and hypothalamus. As a result, pain may be accompanied by emotional distress (suffering) and a variety of autonomic reactions, such as nausea, vomiting, or sweating.
Watch the video below for more information.
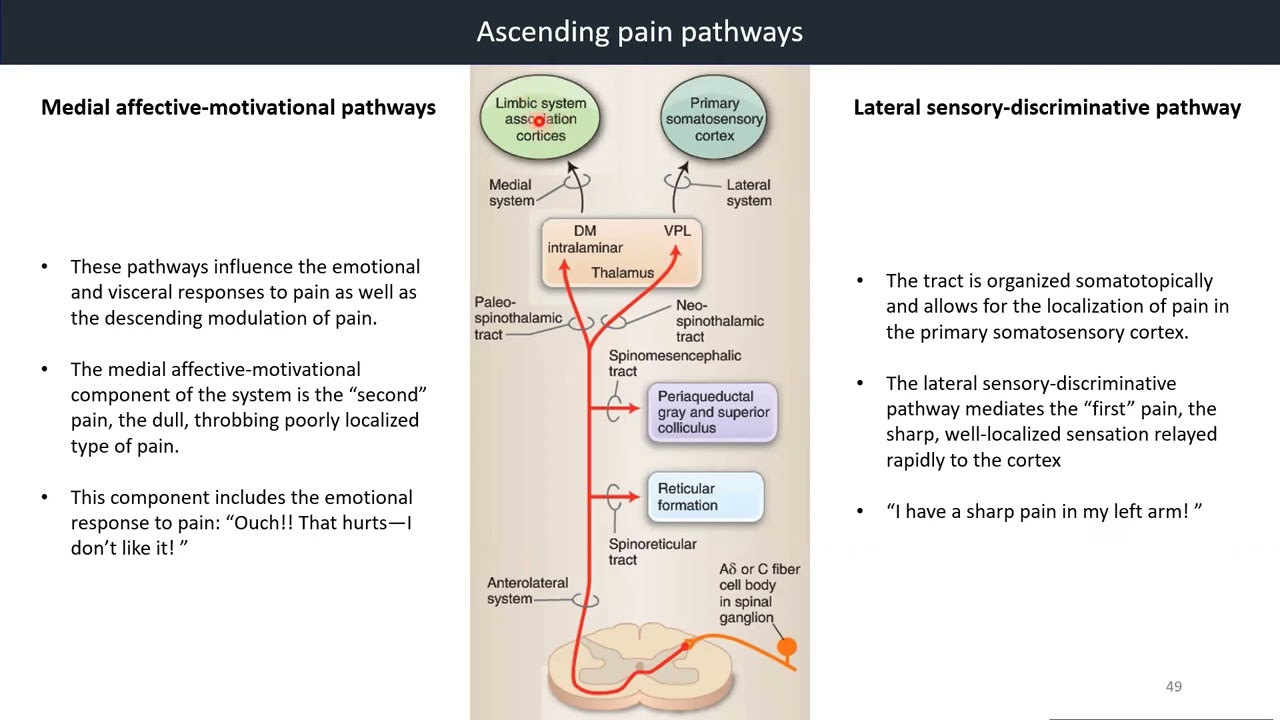
Reference: Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, Figure 22.5
4.9. Gate Control Model of Pain Modulation
Nonpainful stimuli can diminish the pain signal
Pain can also be suppressed in the dorsal horn of the spinal cord, before the stimuli are sent to ascending spinal tracts. Normally, tonically active inhibitory interneurons in the spinal cord inhibit ascending pathways for pain.

C fibers from nociceptors synapse on these inhibitory interneurons. When activated by a painful stimulus, the C fibers simultaneously excite the ascending path and block the tonic inhibition. This action allows the pain signal from the C fiber to travel unimpeded to the brain.

In the gate control theory of pain modulation, A-beta fibers carrying sensory information about mechanical stimuli help block pain transmission. The A-beta fibers synapse on the inhibitory interneurons and enhance the interneuron's inhibitory activity. If simultaneous stimuli reach the inhibitory neuron from the A-beta and C fibers, the integrated response is partial inhibition of the ascending pain pathway so that pain perceived by the brain is lessened.
The gate control theory explains why rubbing a bumped elbow or shin lessens your pain: the tactile stimulus of rubbing activates A-beta fibers and helps decrease the sensation of pain.
4.10. Conceptual Overview of Pain Modulation Network
Descending influences come from the brain stem
Other modulatory systems come from a number of areas in the brainstem, which receive input from the ascending nociceptive pathways.
Video 20: Conceptual Overview of Pain Modulation Network
Watch the video below for more information

Reference: (1) Bears et al., Neuroscience: exploring the brain, 4th edition, Figure 12.34; (2) Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, Figure 22.8
4.11. Endogenous Opioid System
Video 5: Labeled Lines
- Endogenous opioid system provides modulatory influence on cortical pain process.
- Opioid receptors can be found at all levels of the pain system. Opioid receptors are physiologically activated by a group of endogenous molecules that comprise enkephalins, endorphins, and dynorphins. These opioid peptides act as neurotransmitters or neuromodulators and can produce potent analgesic effects.
- Activation of opioid receptors results in the inhibition of voltage-gated Ca2+ channels and/or the opening of K+ channels, which results in hyperpolarization and less neuronal excitability.
- Placebo effect
4.12. Four Main Types of Pain

Pain distributed over an area that is consistent with the boundaries of a dermatome.

Pain that is perceived in a surface area of the body far removed from its actual source.
Pain that is felt in a part of the body that either no longer exists because of amputation or is insensate caused by nerve severance.

Pain arises from a lesion in the thalamus or cortex that is interpreted as pain in the body part corresponding to the lesion.
4.13. Summary: Pain
4.14. Further Readings
-
- Bears et al., Neuroscience: exploring the brain, 4th edition, chapter 8, 11 and 12
- Krebs et al., Lippincott’s Illustrated Reviews: Neuroscience, chapter 22
- Silverthorn, Human Physiology, 5th edition, chapter 10
